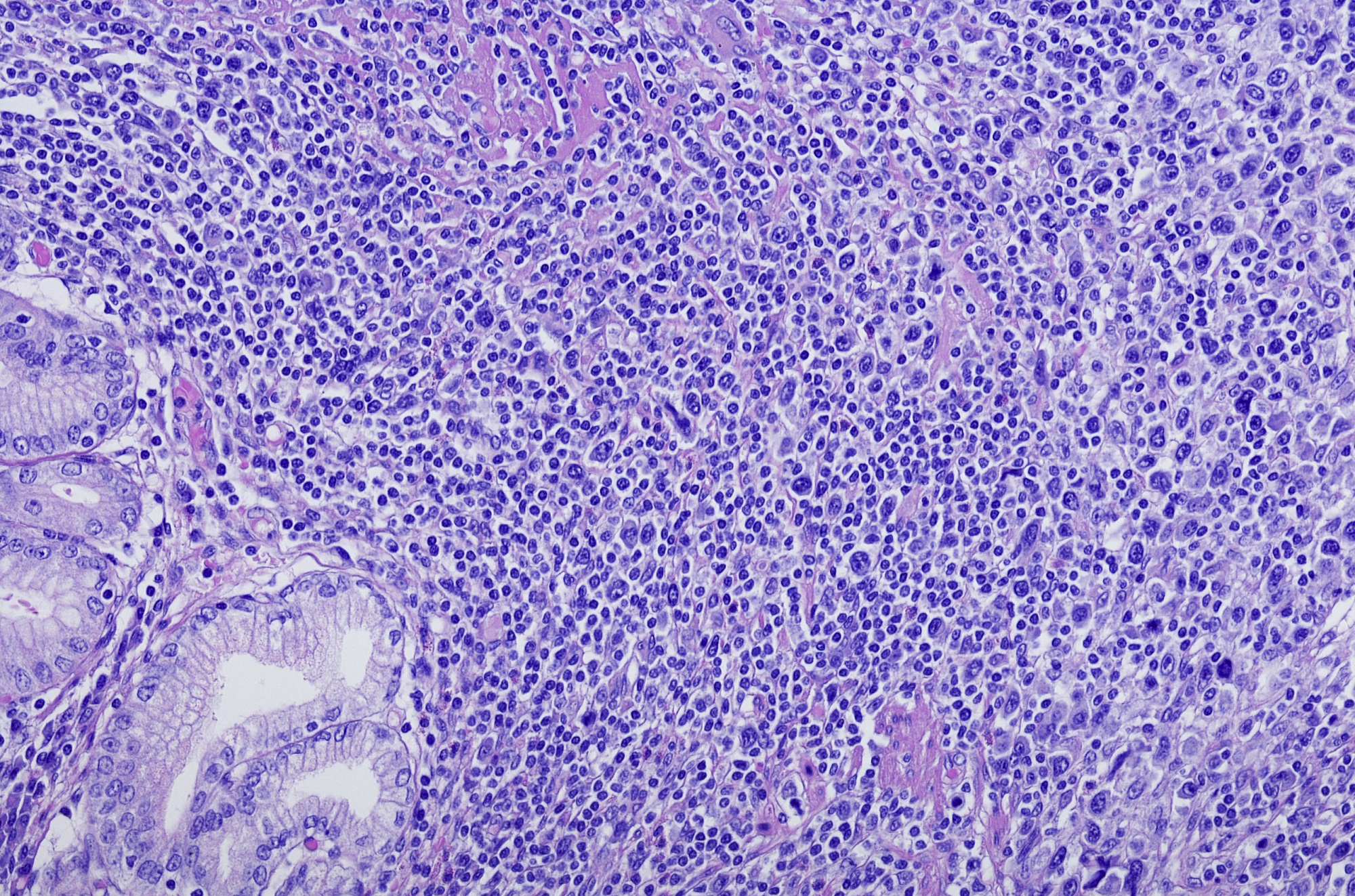

Axicabtagene ciloleucel (axi-cel) appears to be both safe and effective for treating relapsed/refractory follicular lymphoma (FL), according to research presented at the European Hematology Association (EHA) 2021 Virtual Congress.
The phase 2 ZUMA-5 trial (ClinicalTrials.gov Identifier: NCT03105336) previously demonstrated that axi-cel, a chimeric antigen receptor (CAR)-T cell therapy targeting CD19, showed a both high overall response rate and long duration of response in patients with relapsed/refractory FL, including in patients with high-risk disease.
In this presentation, the authors reported comparative clinical outcomes from updated ZUMA-5 data and the international SCHOLAR-5 cohort.
Data from the SCHOLAR-5 cohort were taken from 7 institutions, and included data from a previous trial of idelalisib; patients in SCHOLAR-5 had received a third-line or later therapy after July 2014. Patient characteristics were balanced between the ZUMA-5 and SCHOLAR-5 cohorts.
Overall, data from 86 patients in ZUMA-5 and 85 patients in SCHOLAR-5 were included; the median follow-up times were 23.3 months and 26.2 months, respectively. Patients in the SCHOLAR-5 cohort had more patients with an Eastern Cooperative Oncology Group score of 1 at baseline, despite attempts to match characteristics.
In patients who had received at least 2 prior therapy lines, the overall response rates were 49.9% in SCHOLAR-5 vs 94.2% in ZUMA-5 (odds ratio, 16.2; 95% CI, 5.6-46.9). The median progression-free survival and overall survival periods were not reached in ZUMA-5, and were 12.7 months and 59.8 months, respectively, in SCHOLAR-5; the corresponding hazard ratios for progression-free and overall survival were 0.30 (95% CI, 0.18-0.49) and 0.42 (95% CI, 0.21-0.83), respectively.
“These data support that axi-cel represents a significant improvement in treatment options for patients with relapsed/refractory FL,” the presenter said.
Disclosure: The presenter declared affiliations with AstraZeneca, Kite, A Gilead Company, Gilead, Abbvie, Bristol Myers Squibb, MorphoSys, Novartis, Takeda, TG Therapeutics, Janssen.
Reference
Ghione P, Anik Patel A, Bobillo S, et al. A comparison of clinical outcomes from ZUMA-5 (axicabtagene ciloleucel) and the international SCHOLAR-5 external control cohort in relapsed/refractory follicular lymphoma (R/R FL). Paper presented at: European Hematology Association 2021 Virtual Congress; June 2021; Abstract LB1904.
The post Relapsed, Refractory Follicular Lymphoma: CAR-T Cell Therapy Shows Promise appeared first on Cancer Therapy Advisor.
more recommended stories
 Breast Cancer Risk Rises with Aging Tissue Changes
Breast Cancer Risk Rises with Aging Tissue ChangesKey Highlights A 3-million-cell atlas reveals.
 Type 2 Diabetes Risk Rising in Genetically Susceptible
Type 2 Diabetes Risk Rising in Genetically SusceptibleQuick Summary Rising type 2 diabetes.
 Female Microbiome Shaped by Diet, Stress, Unhealthy Lifestyle
Female Microbiome Shaped by Diet, Stress, Unhealthy LifestyleQuick Summary Lifestyle factors significantly influence.
 Transcatheter Valve-in-Valve Improves Mitral Outcomes
Transcatheter Valve-in-Valve Improves Mitral OutcomesKey Highlights Transcatheter mitral valve-in-valve (mVIV).
 Stroke Rehabilitation: Early High-Intensity Therapy Findings
Stroke Rehabilitation: Early High-Intensity Therapy FindingsKey Highlights High-intensity therapy within 2.
 TRPM8 Cold Sensation Mechanism Explained for Pain Care
TRPM8 Cold Sensation Mechanism Explained for Pain CareQuick Summary TRPM8 ion channel converts.
 Gum Recession from Snus Confirmed, Caries Risk Debated
Gum Recession from Snus Confirmed, Caries Risk DebatedKey Highlights Snus use is strongly.
 Hypertensive Disorders of Pregnancy: Role of Daily Activity
Hypertensive Disorders of Pregnancy: Role of Daily ActivityKey Points Summary Limiting sedentary time.
 Climate Change Drives Dengue Outbreaks Globally
Climate Change Drives Dengue Outbreaks GloballyKey Takeaways Extreme weather significantly increases.
 Teen Driving Risks: Parents Underestimate Safety Threats
Teen Driving Risks: Parents Underestimate Safety ThreatsKey Takeaways Teen driving risks remain.

Leave a Comment