
Key Takeaways for Clinicians
- Chronic hypoxia lowers fasting glucose and improves glucose tolerance independent of insulin
- Red Blood Cells (RBCs) act as a major glucose sink under low-oxygen conditions
- Hypoxia-driven erythrocytosis and metabolic rewiring enhance systemic glucose disposal
- Findings open potential therapeutic directions for type 1 and type 2 diabetes
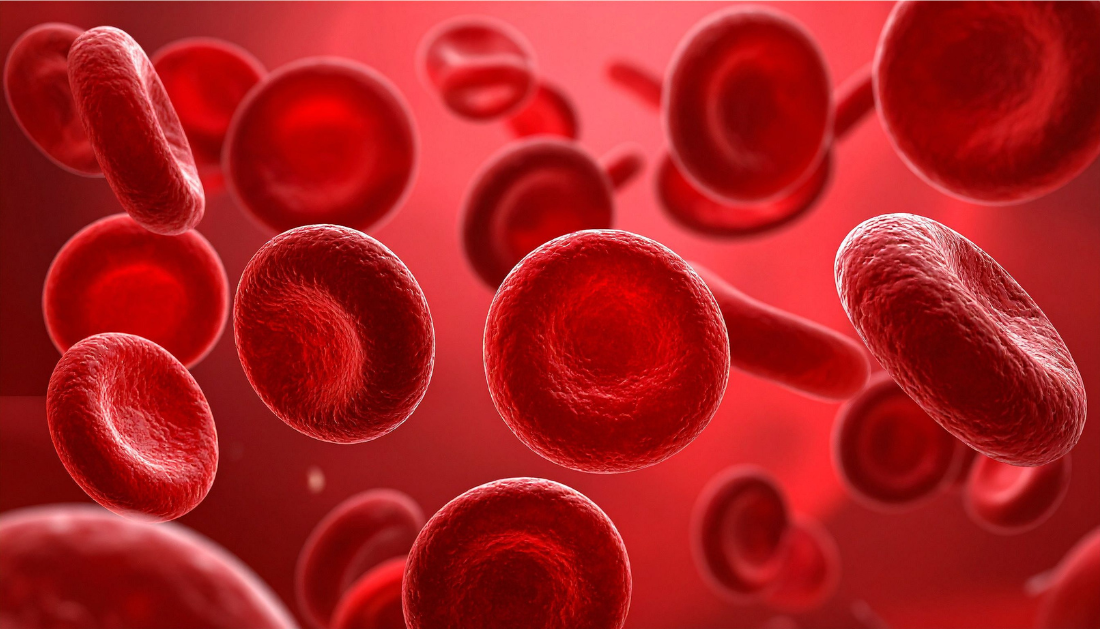
How Red Blood Cells Improve Glucose Tolerance
Populations living at high altitude consistently show lower diabetes prevalence and better glucose control despite reduced oxygen availability. A new study published in Cell Metabolism provides mechanistic clarity, identifying red blood cells as active regulators of systemic glucose metabolism during chronic hypoxia. This work challenges the long-held view of RBCs as passive oxygen carriers and highlights their metabolic relevance in glycemic control.
Hypoxia Rewires Red Blood Cell Glucose Handling
Using a normobaric hypoxia mouse model, researchers demonstrated that exposure to low oxygen rapidly reduced fasting blood glucose and improved glucose tolerance within days. Notably, these effects persisted even after return to normoxia and occurred despite no improvement in insulin sensitivity, suggesting an insulin-independent mechanism.
Chronic hypoxia nearly doubled circulating RBC counts. When hypoxia-induced erythrocytosis was reversed through serial phlebotomy, glycemic benefits disappeared. Conversely, transfusion of RBCs from hypoxic donors into normoxic mice induced hypoglycemia. These findings confirmed that increased RBC mass was both necessary and sufficient for improved glucose tolerance.
Beyond quantity, hypoxic RBCs showed higher per-cell glucose uptake. Flow cytometry revealed increased expression of GLUT1 and GLUT4 transporters, particularly in newly generated erythrocytes. Stable isotope tracing further confirmed accelerated intracellular glucose phosphorylation, indicating enhanced metabolic capacity at the cellular level.
Implications for Diabetes Biology and Care
Metabolomic analyses showed that hypoxic RBCs redirected glucose toward the Luebering–Rapoport shunt, increasing 2,3-diphosphoglycerate production. This pathway not only boosts oxygen delivery to tissues but also increases glucose consumption. Molecular studies identified displacement of GAPDH from band 3 protein under low oxygen as a key trigger for increased glycolytic flux.
Therapeutically, hypoxia exposure and hypoxic RBC transfusion improved hyperglycemia in mouse models of both type 1 and type 2 diabetes. In diet-induced diabetes, pharmacologic induction of tissue hypoxia improved glycemia without direct RBC transfusion, highlighting translational potential.
Why This Matters for HCPs
Explore All Endocrinology CME Conferences & Online Courses
This study positions RBCs as dynamic participants in glucose homeostasis. Targeting erythrocyte metabolism or safely mimicking hypoxia-induced adaptations may offer novel strategies for managing hyperglycemia, especially in insulin-deficient or insulin-resistant states.
Source:
more recommended stories
 Early Autism Detection Using Wearable Sensors in Infants
Early Autism Detection Using Wearable Sensors in InfantsKey Highlights: Wearable movement sensors may.
 Alzheimer’s Disease Driven by Cancer Gene Mutations
Alzheimer’s Disease Driven by Cancer Gene MutationsKey Highlights Microglial cells in Alzheimer’s.
 Cardiovascular Risk Rises with Heatwaves Over 38°C
Cardiovascular Risk Rises with Heatwaves Over 38°CKey Highlights Extreme heat (>38°C) significantly.
 Psychedelic Use Linked to Major Life Changes Study
Psychedelic Use Linked to Major Life Changes StudyQuick Summary 83% of users reported.
 Nutrition Research: Why Context Matters for Healthy Foods
Nutrition Research: Why Context Matters for Healthy FoodsQuick Summary Foods do not have.
 Hyperemesis Gravidarum Genes: Largest GWAS Study
Hyperemesis Gravidarum Genes: Largest GWAS StudyQuick Summary Largest GWAS identifies 10.
 Brain Categorization Redefined by New Neuroscience Study
Brain Categorization Redefined by New Neuroscience StudyKey Highlights Brain Categorization is not.
 GLP-1 Drug Effectiveness Reduced by Genetic Variants
GLP-1 Drug Effectiveness Reduced by Genetic VariantsKey Points Summary Genetic variants in.
 TB Vaccines Show Safety but Limited Protection in Large Trial
TB Vaccines Show Safety but Limited Protection in Large TrialKey Highlights Two candidate Tuberculosis vaccines,.
 Drug Overdose Spike 2025 Was a Modeling Error
Drug Overdose Spike 2025 Was a Modeling ErrorQuick Summary A reported U.S. drug.

Leave a Comment