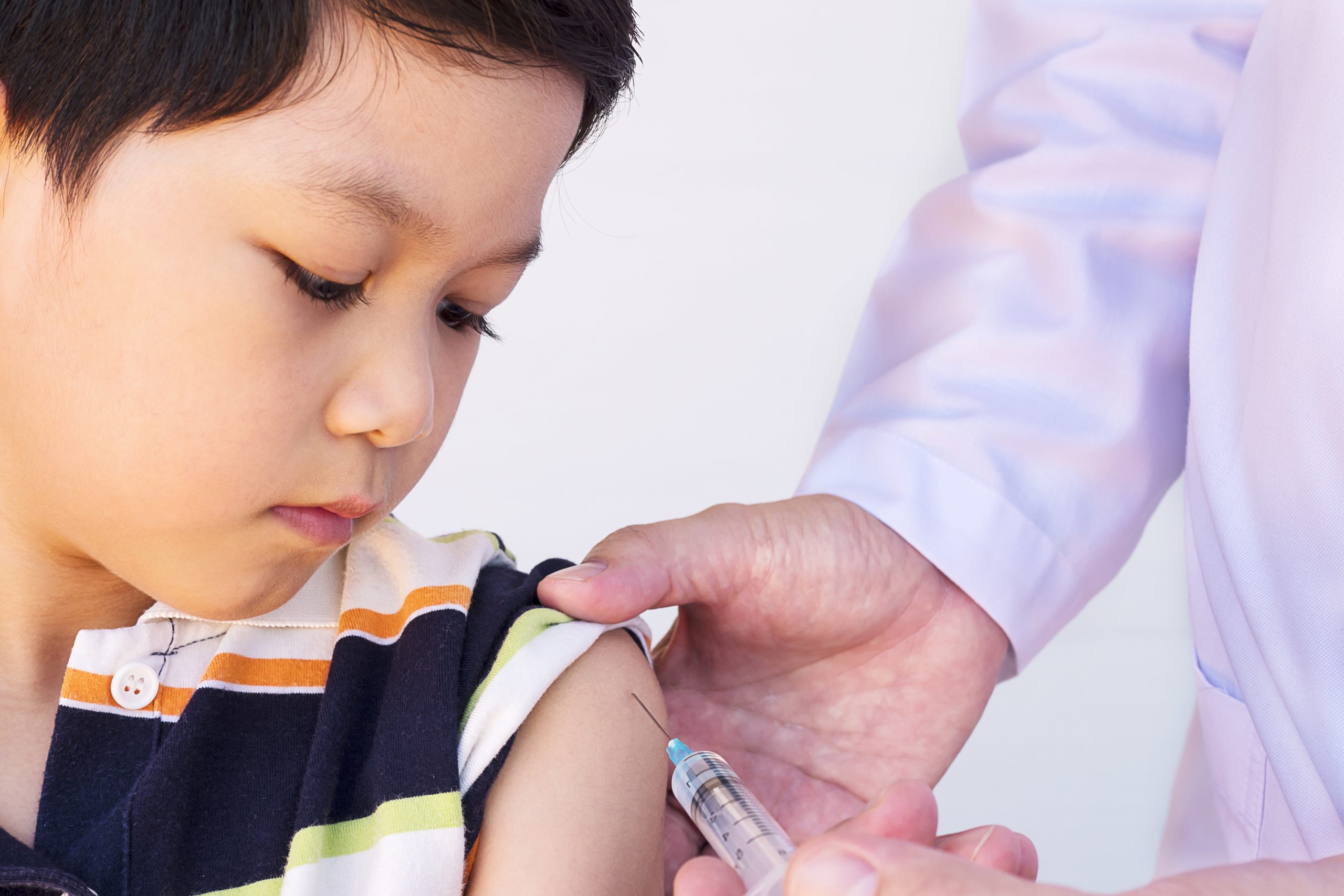

A recent study shows that children with type 1 diabetes between the ages of 2 and 6 benefit from the use of an artificial pancreas that was first created at the University of Virginia Center for Diabetes Technologies. The New England Journal of Medicine has published the specifics of the clinical research as well as its conclusions.
Comparing trial participants utilizing the artificial pancreas to participants in a control group who continued to rely on the methods they were currently using to manage their blood sugar, the artificial pancreas group spent almost three more hours per day in their target blood sugar range.
An automated blood glucose monitor and controller, the Tandem Diabetes Care Control-IQ system is a diabetes management tool.
The artificial pancreas features an insulin pump that can regulate the insulin dose based on data from the patient’s glucose monitoring device. The approach has already been accepted by the U.S. Food and Drug Administration for persons with type 1 diabetes ages 6 and older based on findings from two earlier trials.
“After the resounding success of Control-IQ technology in people ages 6 and up, it is very rewarding to see our youngest patients, and often the most challenging patients to help, benefit as well,” said Marc D. Breton, Ph.D., a UVA School of Medicine researcher who served as the trial’s principal investigator and was recently honored as UVA’s 2022 Innovator of the Year. “With these results, we have now accumulated years of clinical validation of this system across all age groups and look forward to seeing this life-changing technology made available to the broadest possible population.”
102 kids between the ages of 2 and 6 were enrolled in the study at UVA, Stanford University, and the University of Colorado, and 68 of them were randomly assigned to utilize an artificial pancreas system for 13 weeks while the other 34 kids were placed in the control group. Throughout the study, every participant continued to go about their daily lives as usual.
Within their target blood glucose range, participants using the artificial pancreas spent on average roughly 12 percentage points more time than those in the control group did overall, and 18 percentage points more time between the hours of 10 p.m. and 6 a.m. Controlling blood sugar levels at night is crucial because severe, untreated hypoglycemia (extremely low blood sugar levels) can cause seizures, coma, or even death.
Participants were able to utilize the artificial pancreas safely overall, the researchers discovered. In the artificial pancreas group, there were two incidences of severe hypoglycemia, compared to one in the control group. In the artificial pancreas group, there was also one instance of diabetic ketoacidosis brought on by a broken thin plastic line connecting the insulin pump to the patient’s body.
What’s noteworthy is that more than 90% of the visits for the trial were virtual, including 80% of the training sessions for the artificial pancreas. The technology’s simplicity of usage and its promise for regions without convenient access to endocrinologists are highlighted by the stated outcomes being achieved under these circumstances.
“At the end of the day, this technology significantly improved glycemia and ensured the safety of our youngest patients, but perhaps just as importantly it lessened these families’ constant anxiety about glucose levels, especially during the night,” Breton said. “It is incredibly rewarding for us to hear about these families’ experiences and how they manage to integrate these new tools in their life, offering some reprieve to the challenges they face.”
The study’s authors are R. Paul Wadwa, Zachariah W. Reed, Bruce A. Buckingham, Mark D. DeBoer, Laya Ekhlaspour, Gregory P. Forlenza, Melissa Schoelwer, John Lum, Craig Kollman, Roy W. Beck, and Breton.
more recommended stories
 Phage Therapy Study Reveals RNA-Based Infection Control
Phage Therapy Study Reveals RNA-Based Infection ControlKey Takeaways (Quick Summary) Researchers uncovered.
 Safer Allogeneic Stem Cell Transplants with Treg Therapy
Safer Allogeneic Stem Cell Transplants with Treg TherapyA new preclinical study from the.
 AI in Emergency Medicine and Clinician Decision Accuracy
AI in Emergency Medicine and Clinician Decision AccuracyEmergency teams rely on rapid, accurate.
 Innovative AI Boosts Epilepsy Seizure Prediction by 44%
Innovative AI Boosts Epilepsy Seizure Prediction by 44%Transforming Seizure Prediction in Epilepsy Seizure.
 Hypnosis Boosts NIV Tolerance in Respiratory Failure
Hypnosis Boosts NIV Tolerance in Respiratory FailureA New Approach: Hypnosis Improves NIV.
 Bee-Sting Microneedle Patch for Painless Drug Delivery
Bee-Sting Microneedle Patch for Painless Drug DeliveryMicroneedle Patch: A Pain-Free Alternative for.
 AI Reshapes Anticoagulation in Atrial Fibrillation Care
AI Reshapes Anticoagulation in Atrial Fibrillation CareUnderstanding the Challenge of Atrial Fibrillation.
 Hemoglobin as Brain Antioxidant in Neurodegenerative Disease
Hemoglobin as Brain Antioxidant in Neurodegenerative DiseaseUncovering the Brain’s Own Defense Against.
 Global Data Resource for Progressive MS Research (Multiple Sclerosis)
Global Data Resource for Progressive MS Research (Multiple Sclerosis)The International Progressive MS Alliance has.
 AI Diabetes Risk Detection: Early T2D Prediction
AI Diabetes Risk Detection: Early T2D PredictionA new frontier in early diabetes.

Leave a Comment