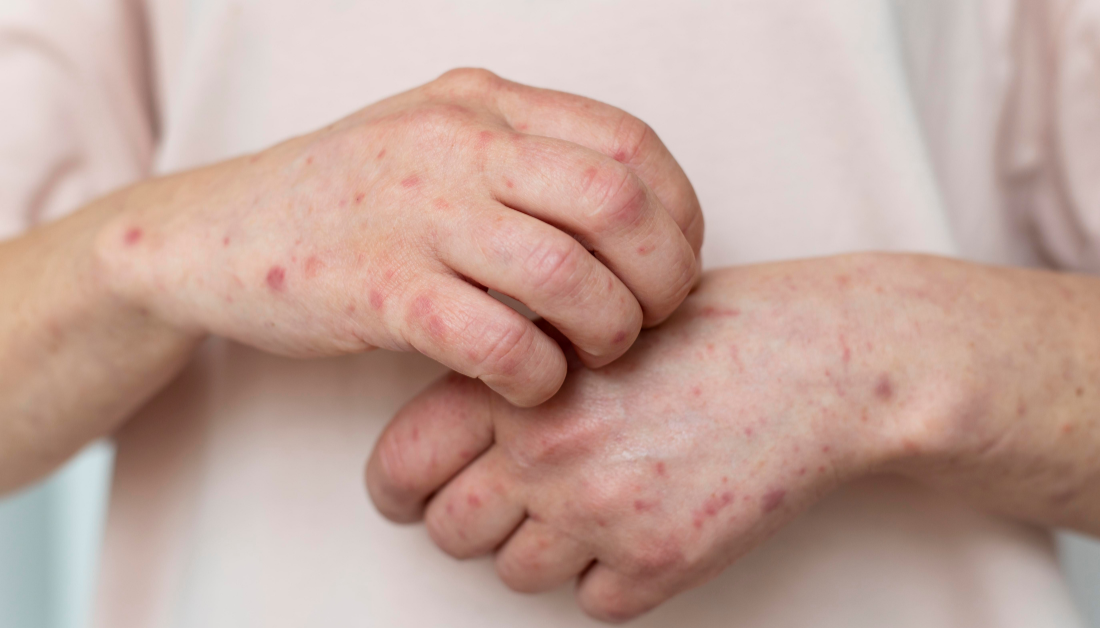

Berdazimer gel 10.3% was approved by the Food and Drug Administration (FDA) on January 5 for the treatment of molluscum contagiosum (MC) in adults and children aged 1 year and older.
Berdazimer, a topical nitric oxide-releasing agent, was approved largely on the basis of B-SIMPLE4, a 12-week pivotal phase 3 trial in which 891 patients with a mean age of 6.6 years (range, 0.9-47.5 years) were randomly assigned to treatment with berdazimer gel 10.3% or a vehicle gel applied in a thin layer to all lesions once daily. At 12 weeks, 32.4% of patients in the berdazimer group had completely cleared MC lesions, compared to 19.7% in the vehicle group (P.001).
Only 4.1% of berdazimer patients and 0.7% of vehicle patients suffered adverse events that required therapy cessation. Both groups had the most prevalent adverse effects, which were mostly mild or severe application-site discomfort and erythema.
According to a press release issued by Ligand Pharmaceuticals, which acquired berdazimer topical gel from Novan in September 2023, the approval makes berdazimer topical gel 10.3% the first and only topical prescription medication that can be used to treat MC at home, outside of a physician’s office, or outside of other medical settings. Although the mechanism of action of berdazimer for treating molluscum “is unknown,” it has been proven to have antiviral properties.
The medication will be branded as Zelsuvmi and is scheduled to be accessible in the second half of 2024.
On July 21, 2023, the FDA approved a drug-device combination (Ycanth) that contains a formulation of cantharidin solution 0.7% and is administered by healthcare professionals, making topical cantharidin the first approved treatment of MC for adults and pediatric patients aged 2 years or older.
For more information: Novel Drug Approvals for 2024
more recommended stories
 Stem Cell Therapies Course for Parkinson’s Disease Clinicians
Stem Cell Therapies Course for Parkinson’s Disease CliniciansKey Summary The International Society for.
 Food Tolerance Mechanism: How T Cells Prevent Allergies
Food Tolerance Mechanism: How T Cells Prevent AllergiesKey Summary Researchers at Stanford University.
 Fragmentome Technology Detects Early Liver Fibrosis
Fragmentome Technology Detects Early Liver FibrosisKey Points at a Glance AI-based.
 AI Predicts Chronic GVHD Risk After Stem Cell Transplant
AI Predicts Chronic GVHD Risk After Stem Cell TransplantKey Takeaways A new AI-driven tool,.
 Rising Measles Cases Prompt Vaccination Push in NC
Rising Measles Cases Prompt Vaccination Push in NCKey Highlights 15 confirmed Measles cases.
 High-Fat Diets Cause Damage to Metabolic Health
High-Fat Diets Cause Damage to Metabolic HealthKey Points Takeaways High-fat and ketogenic.
 Chronic Brain Compression Triggers Neuron Death Pathways
Chronic Brain Compression Triggers Neuron Death PathwaysKey Takeaways Chronic brain compression directly.
 Texas Medical Board Releases Abortion Training for Physicians
Texas Medical Board Releases Abortion Training for PhysiciansKey Takeaways Texas Medical Board has.
 Needle-Thin Brain Implant for Layer-Specific Brain Research
Needle-Thin Brain Implant for Layer-Specific Brain ResearchKey Takeaways Researchers have developed a.
 Pancreatic Cancer Research: Triple-Drug Therapy Success
Pancreatic Cancer Research: Triple-Drug Therapy SuccessKey Summary Spanish researchers report complete.

Leave a Comment